GMP Biologics Market Industry Analysis and Forecast (2026-2032)
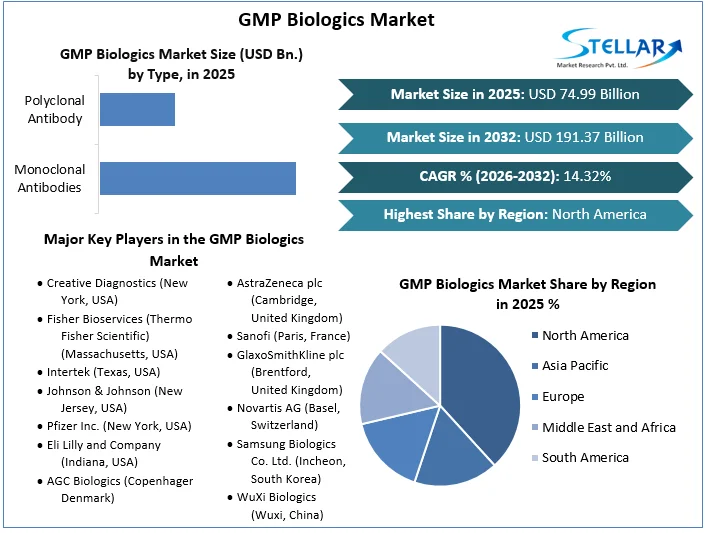
The GMP Biologics Market size was valued at USD 74.99 Bn. in 2025 and the total GMP Biologics Market revenue is expected to grow at a CAGR of 14.32% from 2026 to 2032, reaching nearly USD 191.37 Bn.
GMP Biologics Market Overview
Good Manufacturing Practice (GMP) Biologics refers to the production of biologic medical products under stringent regulatory guidelines designed to ensure product quality, safety, and efficacy. GMP standards, as outlined by organizations like the World Health Organization (WHO), mandate that biologic manufacturing processes are controlled and documented, minimizing risks such as contamination and ensuring consistency across production batches.
North America dominates the GMP Biologics market, owing to its advanced healthcare infrastructure, high healthcare expenditure, and presence of leading biopharmaceutical companies such as Pfizer, and Johnson & Johnson. The region’s dominance is also supported by a strong regulatory framework that facilitates the approval and commercialization of biologic drugs. Europe also holds a significant share of the market, with countries like Germany, the UK, and France leading in terms of R&D investments and manufacturing capabilities.
The increasing adoption of monoclonal antibodies and biosimilars, technological advancements in bioprocessing, and a growing focus on personalized medicine are the key trends are shaping the GMP Biologics market. Monoclonal antibodies, in particular, are becoming increasingly important in the treatment of various diseases, including cancer and autoimmune disorders, due to their high specificity and efficacy. The development of biosimilars is providing more cost-effective treatment options, driving market growth.
Opportunities in the GMP Biologics market are abundant, particularly in emerging markets where healthcare infrastructure is rapidly improving, and there is a high unmet need for advanced biologic therapies. Companies are increasingly focusing on these regions to expand their market presence and tap into the growing demand for biologic drugs. Moreover, advancements in bioprocessing technologies are enabling more efficient and cost-effective production of biologics, further enhancing market growth.
Recent developments by key market players highlight the dynamic nature of the GMP Biologics market. For instance, WuXi Biologics, a leading global open-access biologics technology platform company, continues to expand its capabilities and capacity to meet the growing demand for biologic drugs. The company has been actively investing in new facilities and technologies to enhance its end-to-end solutions for biologics development and manufacturing.
To get more Insights: Request Free Sample Report
GMP Biologics Market Dynamics:
Investment in Advanced Manufacturing Facilities drive the market growth:
Substantial investments in cutting-edge manufacturing facilities are a crucial driver of GMP Biologics Market growth. For instance, Leanbio’s additional €20 million investment in its Barcelona GMP biologics plant highlights its commitment to expanding production capacity and enhancing technological capabilities. This new facility will house production lines for recombinant proteins, antibodies, plasmid DNA, and mRNA, reinforcing Europe’s position as a leader in biopharmaceutical manufacturing and meeting the growing global demand for biologics.
Companies like Bionova Scientific are expanding their manufacturing capacities to meet increasing customer demands. Bionova, a subsidiary of Asahi Kasei Group, has decided to quadruple its GMP manufacturing capacity to support the process development and production of complex next-generation antibody-based drugs. This growth will cater to the growing biopharmaceutical market and leverage Bionova's expertise in managing difficult manufacturing processes.
Partnerships between companies create integrated service offerings that enhance GMP Biologics Market opportunities. Reig Jofre’s investment in Leanbio exemplifies such strategic collaborations. By increasing its stake and integrating services for the development, characterization, and production of biotechnological active ingredients, Reig Jofre offer comprehensive solutions from development to commercialization. This collaboration supports the growth of capabilities and GMP Biologics Market reach, catering to both multinational corporations and start-ups in Europe.
Technological Innovations creating opportunity for the market growth:
Technological advancements in manufacturing processes are setting new standards in the industry. The Boston Institute of Biotechnology (BIB) completed a groundbreaking 30,000L fermentation run, marking a significant milestone in biotechnology. This achievement not only showcases the ability to scale up production efficiently but also demonstrates how innovations in fermentation processes drive GMP Biologics Market growth by increasing production capacity and maintaining high performance standards.
Regulatory advancements, such as the progress in Biologics License Applications (BLA), are vital for GMP Biologics Market growth. For example, Abeona Therapeutics’ ongoing review process for its BLA for prademagene zamikeracel (pz-cel) demonstrates the importance of regulatory milestones in bringing new biologics to market. Successful inspections and mid-cycle reviews by the FDA, without any significant observations, underscore the quality and compliance of manufacturing practices, which is crucial for gaining GMP Biologics Market approval and fostering growth.
The increasing demand for advanced biologics, such as monoclonal antibodies and therapeutic proteins, drives the need for expanded manufacturing capabilities. Aragen’s $30 million investment in a new biologics manufacturing facility in Bangalore, India, is a response to this demand. The facility will feature state-of-the-art equipment for intensified processing and downstream purification, supporting the production of mAbs, therapeutic proteins, and fusion proteins. This growth not only enhances Aragen’s bioproduction capabilities but also meets the global demand for high-quality biologics.
GMP Biologics Market Segment Analysis:
Based on Type, Monoclonal antibodies (mAbs) dominate due to their targeted therapeutic efficacy, which makes them preferable for treating a variety of diseases, particularly in oncology and chronic conditions. This segment's dominance is expected to continue, driven by ongoing advancements in biopharmaceutical research and the increasing demand for personalized medicine. MAbs are widely adopted in hospitals and clinical settings for their precision in targeting specific antigens, which enhances treatment outcomes and minimizes side effects. Polyclonal antibodies, though useful in diagnostics and research due to their ability to recognize multiple epitopes, occupy a smaller GMP Biologics Market share. Their broad reactivity and lower production costs make them valuable in diverse applications, yet their market growth is limited compared to highly specific and effective monoclonal antibodies.
GMP Biologics Market Regional Insights:
North America dominates the market, driven by the presence of numerous well-established biopharmaceutical companies and a robust regulatory framework supporting biologic drug development. Key players in this region include giants like Pfizer, Amgen, and Johnson & Johnson, which leverage advanced technologies and substantial R&D investments to maintain their GMP Biologics Market position. Additionally, the United States has a high adoption rate of biologics in treating chronic diseases and cancer, further propelling GMP Biologics Market growth.
Europe follows closely, supported by a strong biopharmaceutical industry and favorable government policies promoting biotech innovations. Countries like Germany, the UK, and France are leading contributors, with significant investments in biologics R&D and manufacturing capabilities. The European Medicines Agency (EMA) plays a crucial role in facilitating GMP Biologics Market access for new biologics, contributing to the region's strong market presence. Moreover, collaborations and partnerships among European biotech firms and research institutions are enhancing the development and commercialization of novel biologics.
The Asia-Pacific region is expected to witness the highest growth rate in the coming years, primarily due to increasing healthcare expenditure, a rising prevalence of chronic diseases, and improving healthcare infrastructure. Countries like China, India, and South Korea are emerging as key players, driven by supportive government initiatives, expanding biotech industries, and growing investment in biologics research. For instance, China has implemented policies to accelerate drug approvals and increase investments in biopharmaceutical R&D, fostering a conducive environment for biologics market growth. Companies like WuXi Biologics are at the forefront, offering end-to-end solutions and contributing to the region's rapid GMP Biologics Market growth.
GMP Biologics Market Competitive Landscape
Key developments such as investment in a state-of-the-art GMP biologics plant will drive GMP Biologics Market growth by significantly enhancing production capacity for advanced therapies, including recombinant proteins, antibodies, plasmid DNA, and mRNA. This facility, equipped with cutting-edge technology, will support the growing demand for biologics, enable quicker scalability of production, and ensure high-quality standards. These advancements will attract more global projects, strengthen strategic partnerships with major pharmaceutical companies, and position Europe as a leader in biopharmaceutical manufacturing.
On April 25, 2024, the Boston Institute of Biotechnology (BIB), in collaboration with BIBo-Biopharma, achieved a significant milestone in biotechnology by completing a groundbreaking 30,000L fermentation run. This largest-ever capacity GMP run sets a new standard in biologics manufacturing. The team excelled at every stage, maintaining control and performance standards comparable to smaller fermentations. This achievement positions BIB as a pioneer in large-scale upstream primary recovery, underscoring their commitment to innovation and pushing the boundaries of what is possible in the field of biologics manufacturing.
On April 11, 2024, Reig Jofre announced a €5.8 million investment in Leanbio, boosting its stake to 24% in the Barcelona-based biotechnology company. This investment aims to achieve vertical integration in developing and producing biotechnological active ingredients. It will enhance integrated services for recombinant proteins, antibodies, and advanced therapies, benefiting multinational corporations and the startup ecosystem in Spain and Europe. Leanbio will advance its growth by establishing a new GMP-certified plant for industrial-scale production of biotechnological active ingredients and mRNA, further leveraging its expertise in biotechnological product development.
On October 30, 2023, Aragen announced a $30 million investment to establish a new biologics manufacturing facility in Bangalore, India. This state-of-the-art, 160,000 sqm site will feature process development labs, multiple GMP manufacturing suites, and quality control labs. Equipped with single-use bioreactors and advanced purification technology, the facility aims to enhance Aragen's capabilities in developing and manufacturing monoclonal antibodies, therapeutic proteins, and fusion proteins. It will provide comprehensive services from process development to large-scale manufacturing, significantly expanding Aragen's bioproduction capacity alongside its existing California operations.
On December 19, 2023, Leanbio, a global CDMO, announced an additional €20 million investment in its under-construction GMP biologics plant in Sant Quirze del Vallès, Barcelona. This advanced facility, spanning 3,500 sqm, will feature three production lines for recombinant proteins, antibodies, plasmid DNA, and mRNA. Equipped with state-of-the-art technology, it aims to meet the rising demand for biopharmaceutical products, supporting advanced therapies. This growth underscores Leanbio's strategic commitment to enhancing biomanufacturing capabilities and solidifies its position as a key player in the European biologics market.
GMP Biologics Market Scope:
|
GMP Biologics Market |
|
|
Market Size in 2025 |
USD 74.99 Bn. |
|
Market Size in 2032 |
USD 191.37 Bn. |
|
CAGR (2026-2032) |
14.32 % |
|
Historic Data |
2020-2025 |
|
Base Year |
2025 |
|
Forecast Period |
2026-2032 |
|
Segments |
By Type Monoclonal Antibodies Polyclonal Antibody |
|
By Application Hospitals Clinics Others |
|
|
Regional Scope |
North America- United States, Canada, and Mexico Europe – UK, France, Germany, Italy, Spain, Sweden, Russia, and Rest of Europe Asia Pacific – China, India, Japan, South Korea, Australia, ASEAN, Rest of APAC Middle East and Africa - South Africa, GCC, Egypt, Nigeria, Rest of the Middle East and Africa South America – Brazil, Argentina, Rest of South America |
GMP Biologics Market Key Players:
North America:
- Creative Diagnostics (New York, USA)
- Fisher Bioservices (Thermo Fisher Scientific) (Massachusetts, USA)
- Intertek (Texas, USA)
- Johnson & Johnson (New Jersey, USA)
- Pfizer Inc. (New York, USA)
- Eli Lilly and Company (Indiana, USA)
Europe
- AGC Biologics (Copenhagen, Denmark)
- AstraZeneca plc (Cambridge, United Kingdom)
- Sanofi (Paris, France)
- GlaxoSmithKline plc (Brentford, United Kingdom)
- Novartis AG (Basel, Switzerland)
Asia-Pacific
- Samsung Biologics Co. Ltd. (Incheon, South Korea)
- WuXi Biologics (Wuxi, China)
Frequently Asked Questions
Technological Innovations are major opportunity for the GMP Biologics market.
North America is expected to lead the GMP Biologics Market during the forecast period.
The GMP Biologics Market size was valued at USD 74.99 Billion in 2025 and the total GMP Biologics Market revenue is expected to grow at a CAGR of 14.32 % from 2026 to 2032, reaching nearly USD 191.37 Billion.
The segments covered in the GMP Biologics Market report are by Type, Application, and Region.
1. GMP Biologics Market: Research Methodology
1.1. Research Data
1.1.1. Primary Data
1.1.2. Secondary Data
1.2. Market Size Estimation
1.2.1. Bottom-Up Approach
1.2.2. Top-Down Approach
1.3. Market Breakdown and Data Triangulation
1.4. Assumptions
2. GMP Biologics Market: Executive Summary
2.1. Market Overview
2.2. Market Size (2025) and Forecast (2026 – 2032) and Y-O-Y%
2.3. Market Size (USD) and Market Share (%) – By Segments and Regions
3. GMP Biologics Market: Competitive Landscape
3.1. Stellar Competition Matrix
3.2. Key Players Benchmarking
3.2.1. Company Name
3.2.2. Headquarter
3.2.3. Business Segment
3.2.4. End-user Segment
3.2.5. Y-O-Y%
3.2.6. Revenue (2025)
3.2.7. Profit Margin
3.2.8. Market Share
3.2.9. Company Locations
3.3. Market Structure
3.3.1. Market Leaders
3.3.2. Market Followers
3.3.3. Emerging Players
3.4. Consolidation of the Market
3.4.1. Strategic Initiatives and Developments
3.4.2. Mergers and Acquisitions
3.4.3. Collaborations and Partnerships
3.4.4. Product Launches and Innovation
4. GMP Biologics Market: Dynamics
4.1. Market Trends by Region
4.1.1. North America
4.1.2. Europe
4.1.3. Asia Pacific
4.1.4. Middle East and Africa
4.1.5. South America
4.2. Market Drivers
4.3. Market Restraints
4.4. Market Opportunities
4.5. Market Challenges
4.6. PORTER’s Five Forces Analysis
4.6.1. Intensity of the Rivalry
4.6.2. Threat of New Entrants
4.6.3. Bargaining Power of Suppliers
4.6.4. Bargaining Power of Buyers
4.6.5. Threat of Substitutes
4.7. PESTLE Analysis
4.7.1. Political Factors
4.7.2. Economic Factors
4.7.3. Social Factors
4.7.4. Technological Factors
4.7.5. Legal Factors
4.7.6. Environmental Factors
4.8. Technological Roadmap
4.9. Regulatory Landscape
4.9.1. Market Regulation by Region
4.9.1.1. North America
4.9.1.2. Europe
4.9.1.3. Asia Pacific
4.9.1.4. Middle East and Africa
4.9.1.5. South America
4.9.2. Impact of Regulations on Market Dynamics
4.9.3. Government Schemes and Initiatives
5. GMP Biologics Market Size and Forecast by Segments (by Value USD Million)
5.1. GMP Biologics Market Size and Forecast, By Type (2025-2032)
5.1.1. Monoclonal Antibodies
5.1.2. Polyclonal Antibody
5.2. GMP Biologics Market Size and Forecast, By Application (2025-2032)
5.2.1. Hospitals
5.2.2. Clinics
5.2.3. Others
5.3. GMP Biologics Market Size and Forecast, by Region (2025-2032)
5.3.1. North America
5.3.2. Europe
5.3.3. Asia Pacific
5.3.4. Middle East and Africa
5.3.5. South America
6. North America GMP Biologics Market Size and Forecast (by Value USD Million)
6.1. North America GMP Biologics Market Size and Forecast, By Type (2025-2032)
6.1.1. Monoclonal Antibodies
6.1.2. Polyclonal Antibody
6.2. North America GMP Biologics Market Size and Forecast, By Application (2025-2032)
6.2.1. Hospitals
6.2.2. Clinics
6.2.3. Others
6.3. North America GMP Biologics Market Size and Forecast, by Country (2025-2032)
6.3.1. United States
6.3.2. Canada
6.3.3. Mexico
7. Europe GMP Biologics Market Size and Forecast (by Value USD Million)
7.1. Europe GMP Biologics Market Size and Forecast, By Type (2025-2032)
7.2. Europe GMP Biologics Market Size and Forecast, By Application (2025-2032)
7.3. Europe GMP Biologics Market Size and Forecast, by Country (2025-2032)
7.3.1. UK
7.3.2. France
7.3.3. Germany
7.3.4. Italy
7.3.5. Spain
7.3.6. Sweden
7.3.7. Austria
7.3.8. Rest of Europe
8. Asia Pacific GMP Biologics Market Size and Forecast (by Value USD Million)
8.1. Asia Pacific GMP Biologics Market Size and Forecast, By Type (2025-2032)
8.2. Asia Pacific GMP Biologics Market Size and Forecast, By Application (2025-2032)
8.3. Asia Pacific GMP Biologics Market Size and Forecast, by Country (2025-2032)
8.3.1. China
8.3.2. S Korea
8.3.3. Japan
8.3.4. India
8.3.5. Australia
8.3.6. Indonesia
8.3.7. Malaysia
8.3.8. Vietnam
8.3.9. Taiwan
8.3.10. Bangladesh
8.3.11. Pakistan
8.3.12. Rest of Asia Pacific
9. Middle East and Africa GMP Biologics Market Size and Forecast (by Value USD Million)
9.1. Middle East and Africa GMP Biologics Market Size and Forecast, By Type (2025-2032)
9.2. Middle East and Africa GMP Biologics Market Size and Forecast, By Application (2025-2032)
9.3. Middle East and Africa GMP Biologics Market Size and Forecast, by Country (2025-2032)
9.3.1. South Africa
9.3.2. GCC
9.3.3. Egypt
9.3.4. Nigeria
9.3.5. Rest of ME&A
10. South America GMP Biologics Market Size and Forecast (by Value USD Million)
10.1. South America GMP Biologics Market Size and Forecast, By Type (2025-2032)
10.2. South America GMP Biologics Market Size and Forecast, By Application (2025-2032)
10.3. South America GMP Biologics Market Size and Forecast, by Country (2025-2032)
10.3.1. Brazil
10.3.2. Argentina
10.3.3. Rest of South America
11. Company Profile: Key players
11.1. Creative Diagnostics (New York, USA)
11.1.1. Company Overview
11.1.2. Business Portfolio
11.1.3. Financial Overview
11.1.3.1. Total Revenue
11.1.3.2. Segment Revenue
11.1.3.3. Regional Revenue
11.1.4. SWOT Analysis
11.1.5. Strategic Analysis
11.1.6. Recent Developments
11.2. Fisher Bioservices (Thermo Fisher Scientific) (Massachusetts, USA)
11.3. Intertek (Texas, USA)
11.4. Johnson & Johnson (New Jersey, USA)
11.5. Pfizer Inc. (New York, USA)
11.6. Eli Lilly and Company (Indiana, USA)
11.7. AGC Biologics (Copenhagen, Denmark)
11.8. AstraZeneca plc (Cambridge, United Kingdom)
11.9. Sanofi (Paris, France)
11.10. GlaxoSmithKline plc (Brentford, United Kingdom)
11.11. Novartis AG (Basel, Switzerland)
11.12. Samsung Biologics Co. Ltd. (Incheon, South Korea)
11.13. WuXi Biologics (Wuxi, China)
12. Key Findings
13. Industry Recommendations
13.1. Strategic Recommendations
13.2. Future Outlook
















