Cell Culture Media Market Global Industry Analysis and Forecast (2026–2032) – Growth Opportunities Driven by Serum-Free Media Adoption, Biologics Manufacturing, and Cell & Gene Therapies
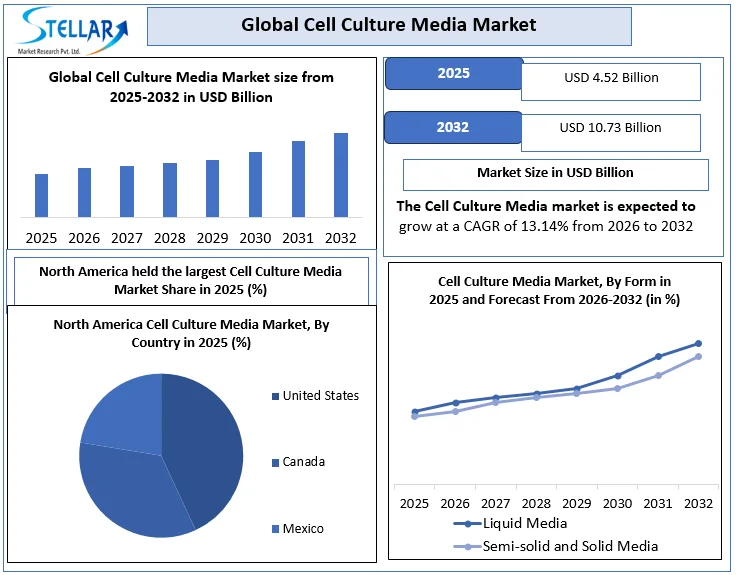
The Global Cell Culture Media Market was valued at USD 4.52 Billion in 2025 and is projected to reach USD 10.73 Billion by 2032, growing at a CAGR of 13.14% during the forecast period (2026–2032). Market growth is driven by rising biologic drug approvals, expansion of cell and gene therapy pipelines, increasing vaccine production, and the rapid shift toward serum-free, chemically defined, and GMP-compliant media formulations.
Cell Culture Media Market Overview:
Cell culture media are nutrient-rich formulations used to support in-vitro cell growth, proliferation, and maintenance across biopharmaceutical production, regenerative medicine, diagnostics, and drug discovery. In 2025, the market is characterized by a structural shift from serum-based media toward serum-free, xeno-free, and chemically defined formulations to ensure reproducibility, scalability, and regulatory compliance.
Advanced media solutions are increasingly deployed in CHO, HEK 293, BHK, and Vero cell-based production systems for monoclonal antibodies, vaccines, and gene therapies. North America leads adoption due to its concentration of biopharmaceutical manufacturers and CDMOs, while Asia Pacific is emerging as a high-growth region driven by expanding biologics capacity.
Key highlights
• In 2025, over 40% of new drug approvals were biologics, with approximately 70% of monoclonal antibodies manufactured using serum-free or chemically defined media.
• North America accounted for nearly 50% of global cell culture media demand in 2025, driven by regenerative medicine, stem cell research, and large-scale biopharmaceutical production.
• Advanced media formulations are 40–60% more expensive than classical media, creating cost pressures for small and mid-sized biotechnology companies.
• Adoption of serum-free media increased by approximately 30% between 2020 and 2025, supported by regulatory requirements and demand for batch consistency.
• More than 80% of biopharmaceutical manufacturers in North America used GMP-compliant cell culture media in 2025.
To get more Insights: Request Free Sample Report
Cell Culture Media Market Trend: Adoption of Serum-Free and Chemically Defined Media
The increasing adoption of serum-free and chemically defined media is driven by the demand for higher consistency and reproducibility in biologic drug production.
- These media eliminate variability from animal-derived components, ensuring more controlled and standardized environments for cell growth.
- Particularly critical in personalized medicine, stem cell therapies, and gene therapies, where maintaining consistent production quality is crucial.
- As regulatory standards for biologic drug production become stricter, the demand for serum-free and chemically defined media continues to surge, with 30% increase in demand over the last five years.
- These media are indispensable in biopharmaceutical manufacturing to meet the growing demand for vaccines, monoclonal antibodies, and other biologic drugs, boosts Cell Culture Media Market growth.
- Standardization and reduction of contamination risks are major factors driving this shift towards serum-free and chemically defined solutions.
Increasing Demand for Biologic Drugs and Therapeutics to Boosts Cell Culture Media Market Growth
The increasing use of monoclonal antibodies, gene therapies, and vaccines all of which require specialized cell culture media for their production.
- In 2025, 70% of monoclonal antibodies were produced using serum-free and chemically defined media, ensuring scalability and consistency.
- Biopharmaceutical manufacturers are shifting to serum-free media, with adoption increasing in 30% from the last five years globally.
- The use of chemically defined media increases by 25% from 2024, as these media eliminate variability and enhance reproducibility in biologic production.
As more treatments shift toward biologic drugs and immunotherapies, the demand for high-quality, scalable, and efficient cell culture media solutions is growing. Biopharmaceutical manufacturers are increasingly adopting advanced media solutions to ensure consistency and regulatory compliance, which boosts the Cell Culture Media Market growth.
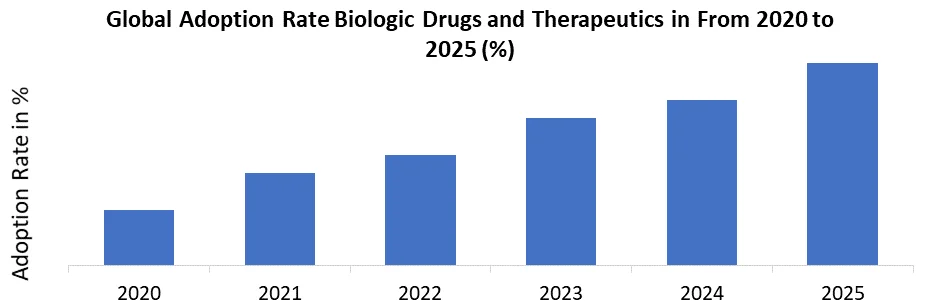
High Cost of Advanced Cell Culture Media to Restraint cell culture media market
The high cost is associated with advanced formulations, such as serum-free and chemically defined media, which hamper the Cell Culture Media Market growth. These advanced media types are more expensive than traditional media, making them less accessible for smaller biotech firms and research labs with limited budgets. The increasing demand for high-quality, reproducible cell growth has led to the development of more specialized media, but this has added to the financial burden faced by many manufacturers.
The continuous need for innovation in developing new, more efficient media formulations adds to costs. Biopharmaceutical companies are investing significant resources into ensuring regulatory compliance, which contributes to the overall price of these media solutions. Although the need for higher-quality media is crucial for biologic drug production, the high upfront investment remains a significant barrier to entry, especially for smaller organizations and early-stage research projects.
Cell Culture Media Market Segment Analysis
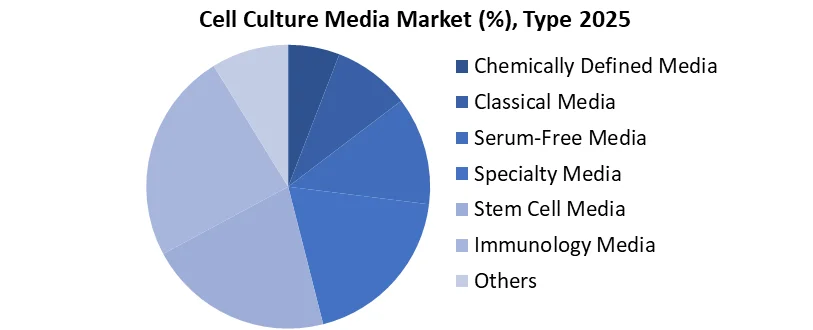
Based on Type, the Cell Culture Media Market is segmented into the Chemically Defined Media, Classical Media, Serum-Free Media, Specialty Media, Stem Cell Media, Immunology Media and Others. Serum-free media is the dominant type in the cell culture media market, experiencing significant growth due to its increasing importance in the production of biopharmaceuticals, particularly for monoclonal antibodies and therapeutic proteins. This media type offers a more consistent and reproducible environment for cell growth, essential for minimizing contamination risks and ensuring high-quality production.
- Regions with even higher adoption rates due to the shift towards animal-free, defined media.
- Essential for ensuring consistency and scalability in production.
- Crucial for manufacturing vaccines, gene therapies, and stem cells.
- Reduces contamination risks and provides reproducible cell growth environments.
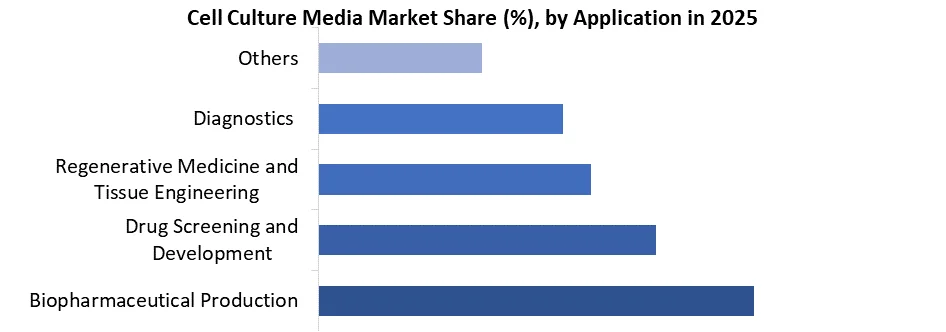
Based on Application, the market is categorized into the Biopharmaceutical Production, Drug Screening and Development, Regenerative Medicine and Tissue Engineering, Diagnostics and Others. The biopharmaceutical production held the largest share for Cell Culture Market in 2025. The increasing demand for biologic drugs such as monoclonal antibodies, vaccines, and gene therapies.
The adoption of serum-free and chemically defined media is expanding rapidly, essential for large-scale production, ensuring consistent and reproducible results critical for regulatory compliance. As of 2025, Over 56% of the total cell culture media market is attributed to biopharmaceutical production, highlighting the sector's dominance. This growth is boosted by the rise of personalized medicine and immune-oncology therapies.
Regional Analysis of the Cell Culture Media Market
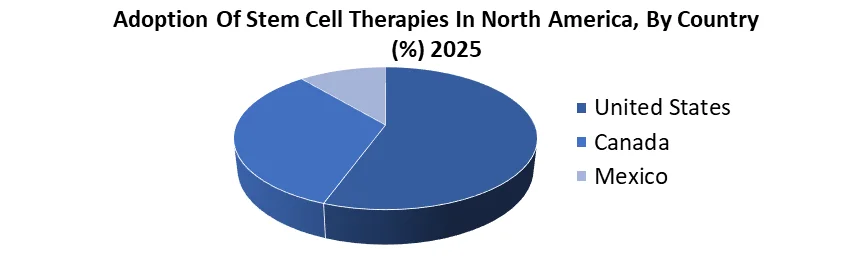
North America held the largest Cell Culture Media Market share in 2025 and is expected to continue its dominance over the forecast period.
- Cell culture media is crucial in advancing regenerative medicine, stem cell research, and biopharmaceutical production in North America.
- Leading suppliers such as Sartorius and PromoCell offer xeno-free, animal-free, and serum-free media, ensuring optimal cell health and reproducibility.
- Custom formulations are available to meet specific research and manufacturing needs, supporting both small-scale and large-scale projects.
- The biopharmaceutical companies in North America use GMP-compliant cell culture media, contributing to market growth.
Cell Culture Media Market Competitive Landscape
The Cell Culture Media Market is highly competitive, with major players such as Thermo Fisher, Merck, Lonza, and Corning leading innovation and expansion. Companies focus on offering serum-free, chemically defined, and specialized media for diverse applications in biopharmaceuticals, diagnostics, and regenerative medicine, while emphasizing quality, scalability, and customization.
Cell Culture Media Market Recent Developments
- September 5, 2024, Merck has achieved EXCiPACT cGMP certification for its global Cell Culture Media (CCM) production sites in Germany, the UK, China, and the USA. This industry-first certification assures the safety, quality, and efficacy of cell culture media used in life-saving therapies like vaccines, gene therapies, and monoclonal antibodies. The EXCiPACT GMP guideline for CCM manufacturing was published in May 2023, reinforcing Merck’s leadership in delivering high-quality, compliant solutions for the biopharma industry.
- April 14, 2025, Sartorius and Mabion announced a strategic collaboration to enhance biopharmaceutical development. Sartorius contributes its proprietary cell culture media and expertise in cell line development, while Mabion focuses on scale-up and large-scale manufacturing. This partnership offers integrated, end-to-end services from lab-scale development to commercial manufacturing, aiming to reduce complexity and accelerate the time to market for biologics.
Global Cell Culture Media Market Scope
|
Global Cell Culture Media Market |
|||
|
Report Coverage |
Details |
||
|
Base Year: |
2025 |
Forecast Period: |
2026-2032 |
|
Historical Data: |
2020 to 2025 |
Market Size in 2025: |
USD 4.52 Billion |
|
Forecast Period 2026 to 2032 CAGR: |
13.14% |
Market Size in 2032: |
USD 10.73 Billion |
|
Cell Culture Media Market Segment Analysis |
By Type |
Chemically Defined Media Classical Media Serum-Free Media CHO Media BHK Medium Vero Medium HEK 293 Media Others Specialty Media Stem Cell Media Mesenchymal Stem Cells (MSCs) Hematopoietic Stem Cells (HSCs) Induced Pluripotent Stem Cells (iPSCs) Others Immunology Media T Cells B Cells Natural Killer (NK) Cells Others Others |
|
|
By Form |
Liquid Media Semi-solid and Solid Media |
||
|
By Application |
Biopharmaceutical Production Monoclonal Antibodies Vaccines Production Others Drug Screening and Development Regenerative Medicine and Tissue Engineering Cell and Gene Therapy Others Diagnostics Others |
||
|
By End User |
Food and Beverage Industry Pharmaceuticals Biotechnology Others |
||
Cell Culture Media Key Players
- Thermo Fisher Scientific Inc.
- Merck KGaA
- Danaher Corporation (includes Cytiva)
- Sartorius AG
- Lonza Group AG
- Corning Incorporated
- Becton Dickinson (BD)
- HiMedia Laboratories Pvt. Ltd.
- Fujifilm Irvine Scientific (FUJIFILM)
- Bio Techne Corporation
- PromoCell GmbH
- CellGenix GmbH
- Takara Bio, Inc.
- ZenBio, Inc.
- Biological Industries Ltd.
- Stemcell Technologies Inc.
- Agilent Technologies, Inc.
- Serumwerk Bernburg AG
- Pan Biotech GmbH
- Atlanta Biologicals, Inc.
- Bio Rad Laboratories, Inc.
- InVitria (Ventria Bioscience)
- Allele Life Sciences (P) Ltd
- PL BioScience GmbH
- Alna Bioscience GmbH & Co. KG
- Biomatik Corporation
- Biologos
- Creative Bioarray
- Cell Culture Technologies
- Quality Biological, Inc.
Frequently Asked Questions
North America region is projected to hold the highest share in the Cell Culture Media market due to growth in biotechnology and pharmaceuticals in the region along with growth in trend of regulatory approvals.
The Cell Culture Media market is driven by its importance in vaccine production. Cell culture technology is used for vaccine production of hepatitis, rotavirus, polio and smallpox.
The forecast period is from 2026 to 2032 which covers the duration of 6 years.
Cell Culture Media market is projected to be at a value of USD 10.73 Billion in 2032.
1. Cell Culture Media Market Introduction
1.1. Study Assumptions and Market Definition
1.2. Scope of the Study
1.3. Executive Summary
2. Global Cell Culture Media Market: Competitive Landscape
2.1. SMR Competition Matrix
2.2. Competitive Landscape
2.3. Key Players Benchmarking
2.3.1. Company Name
2.3.2. Headquarter
2.3.3. Product Portfolio
2.3.4. End-User
2.3.5. Total Company Revenue (2025)
2.3.6. Certifications
2.3.7. Global Presence
2.4. Market Structure
2.4.1. Market Leaders
2.4.2. Market Followers
2.4.3. Emerging Players
2.5. Mergers and Acquisitions Details
2.6. Recent Developments
2.7. Market Positioning & Share Analysis
2.7.1. Company Revenue, Cell Culture Media Revenue, and Market Share (%)
2.7.2. SMR Competitive Positioning
2.8. Strategic Developments & Partnerships
2.8.1. Mergers, acquisitions, and joint ventures
2.8.2. Expansion into emerging markets
2.8.3. Strategic alliances with OEMs or system integrators
2.8.4. Investments in new production facilities
2.8.5. Sustainability initiatives and green product launches
3. Cell Culture Media Market: Dynamics
3.1. Cell Culture Media Market Trends
3.2. Cell Culture Media Market Dynamics
3.2.1. North America
3.2.2. Europe
3.2.3. Asia Pacific
3.2.4. Middle East and Africa
3.2.5. South America
3.3. PORTER’s Five Forces Analysis
3.4. PESTLE Analysis
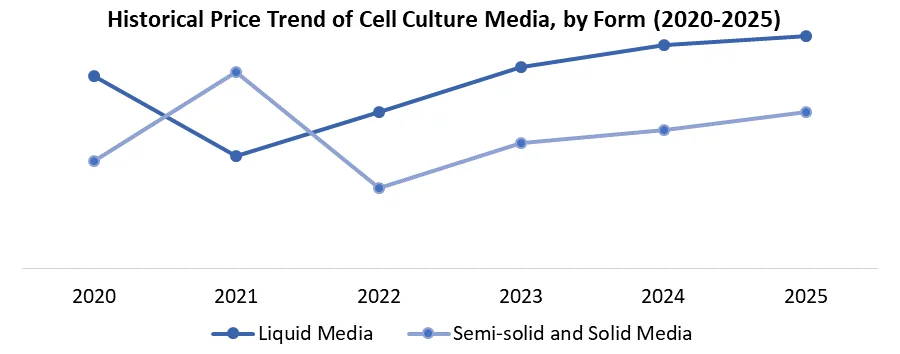
4. Pricing and Cost Structure Analysis
4.1. Price Trend Analysis (2020-2025)
4.2. Factors Influencing Pricing
4.2.1. Regulatory Policies
4.2.2. Market Competition and Market Entry Barriers
4.2.3. Manufacturing and Supply Chain Efficiency
4.3. Cost Breakdown of Advanced Cell Culture Media
5. Adoption and Usage Trends in the Cell Culture Media Market
5.1. Serum-Free and Chemically Defined Media Adoption Trends
5.2. Demand for Biologic Drugs and Therapeutics
5.3. Adoption in Regenerative Medicine and Stem Cell Therapies
5.4. Global Biopharmaceutical Production Growth
5.5. Technology Integration in Cell Culture Media Solutions
6. Government Healthcare Spending and Policies Impact
6.1. National Healthcare Budgets and Policies
6.2. Policy Initiatives Supporting Cell Culture Media Adoption
6.3. Government Funding Allocations Impacting the Biopharmaceutical Sector
6.4. Impact of Public Funding on Cell Culture Media Penetration
7. Patent and Innovation Analysis
7.1. Tracking Loss of Exclusivity (LOE) for Biopharmaceutical Drugs
7.2. Market Entry and Licensing Opportunities for Generic Media Solutions
7.3. Patent Litigation and Regulatory Barriers
7.4. Innovation in Cell Culture Media Formulations
7.5. Trends in Patent Evergreening and Impact on Cell Culture Media Adoption
8. Research and Development Analysis
8.1. Overview of R&D in Cell Culture Media Development
8.2. Investment Trends in R&D for Media Solutions
8.3. Bioequivalence Study Costs and Timelines in Cell Culture Media
8.4. Challenges in Reverse Engineering and Media Formulation
8.5. Focus Areas: Complex Media and Stem Cell-Based Solutions
8.6. Regulatory Support for R&D in Media Solutions
9. Supply Chain Analysis in Cell Culture Media
9.1. Dependency on Raw Material Sourcing (APIs)
9.2. Manufacturing Quality Compliance in Cell Culture Media
9.3. Regulatory Pathways and Licensing Bottlenecks
9.4. Pharmaceutical Distribution and Logistics Challenges
9.5. Sustainability and Green Supply Chain Initiatives
10. EMA-Approved Cell Culture Media Overview
10.1. Number of Cell Culture Media Approvals (2019-2024)
10.2. Therapeutic Area Breakdown of Approved Media
10.3. Efficiency of Regulatory Pathways for Approvals
10.4. Domestic vs. International Manufacturer Approvals
10.5. Time-to-Approval Benchmarks for Various Media Types
11. Cell Culture Media Market: Global Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
11.1. Cell Culture Media Market Size and Forecast, by Type (2025-2032)
11.1.1. Chemically Defined Media
11.1.2. Classical Media
11.1.3. Serum-Free Media
11.1.3.1. CHO Media
11.1.3.2. BHK Medium
11.1.3.3. Vero Medium
11.1.3.4. HEK 293 Media
11.1.3.5. Others
11.1.4. Specialty Media
11.1.5. Stem Cell Media
11.1.5.1. Mesenchymal Stem Cells (MSCs)
11.1.5.2. Hematopoietic Stem Cells (HSCs)
11.1.5.3. Induced Pluripotent Stem Cells (iPSCs)
11.1.5.4. Others
11.1.6. Immunology Media
11.1.6.1. T Cells
11.1.6.2. B Cells
11.1.6.3. Natural Killer (NK) Cells
11.1.6.4. Others
11.1.7. Others
11.2. Cell Culture Media Market Size and Forecast, by Form (2025-2032)
11.2.1. Liquid Media
11.2.2. Semi-solid and Solid Media
11.3. Cell Culture Media Market Size and Forecast, by Application (2025-2032)
11.3.1. Biopharmaceutical Production
11.3.1.1. Monoclonal Antibodies
11.3.1.2. Vaccines Production
11.3.1.3. Others
11.3.2. Drug Screening and Development
11.3.3. Regenerative Medicine and Tissue Engineering
11.3.3.1. Cell and Gene Therapy
11.3.3.2. Others
11.3.4. Diagnostics
11.3.5. Others
11.4. Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
11.4.1. Biotechnology & Pharmaceutical Companies
11.4.2. Research & Academic Institutes
11.4.3. Contract Research & Manufacturing Organizations (CROs/CMOs)
11.4.4. Others
11.5. Cell Culture Media Market Size and Forecast, by Region (2025-2032)
11.5.1. North America
11.5.2. Europe
11.5.3. Asia Pacific
11.5.4. Middle East and Africa
11.5.5. South America
12. North America Cell Culture Media Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
12.1. North America Cell Culture Media Market Size and Forecast, by Type (2025-2032)
12.1.1. Chemically Defined Media
12.1.2. Classical Media
12.1.3. Serum-Free Media
12.1.3.1. CHO Media
12.1.3.2. BHK Medium
12.1.3.3. Vero Medium
12.1.3.4. HEK 293 Media
12.1.3.5. Others
12.1.4. Specialty Media
12.1.5. Stem Cell Media
12.1.5.1. Mesenchymal Stem Cells (MSCs)
12.1.5.2. Hematopoietic Stem Cells (HSCs)
12.1.5.3. Induced Pluripotent Stem Cells (iPSCs)
12.1.5.4. Others
12.1.6. Immunology Media
12.1.6.1. T Cells
12.1.6.2. B Cells
12.1.6.3. Natural Killer (NK) Cells
12.1.6.4. Others
12.1.7. Others
12.2. North America Cell Culture Media Market Size and Forecast, by Form (2025-2032)
12.2.1. Liquid Media
12.2.2. Semi-solid and Solid Media
12.3. North America Cell Culture Media Market Size and Forecast, by Application (2025-2032)
12.3.1. Biopharmaceutical Production
12.3.1.1. Monoclonal Antibodies
12.3.1.2. Vaccines Production
12.3.1.3. Others
12.3.2. Drug Screening and Development
12.3.3. Regenerative Medicine and Tissue Engineering
12.3.3.1. Cell and Gene Therapy
12.3.3.2. Others
12.3.4. Diagnostics
12.3.5. Others
12.4. North America Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
12.4.1. Biotechnology & Pharmaceutical Companies
12.4.2. Research & Academic Institutes
12.4.3. Contract Research & Manufacturing Organizations (CROs/CMOs)
12.4.4. Others
12.5. North America Cell Culture Media Market Size and Forecast, by Country (2025-2032)
12.5.1. United States
12.5.2. Canada
12.5.3. Mexico
13. Europe Cell Culture Media Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
13.1. Europe Cell Culture Media Market Size and Forecast, by Type (2025-2032)
13.2. Europe Cell Culture Media Market Size and Forecast, by Form (2025-2032)
13.3. Europe Cell Culture Media Market Size and Forecast, by Application (2025-2032)
13.4. Europe Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
13.5. Europe Cell Culture Media Market Size and Forecast, by Country (2025-2032)
13.5.1. United Kingdom
13.5.2. France
13.5.3. Germany
13.5.4. Italy
13.5.5. Spain
13.5.6. Sweden
13.5.7. Austria
13.5.8. Rest of Europe
14. Asia Pacific Cell Culture Media Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
14.1. Asia Pacific Cell Culture Media Market Size and Forecast, by Type (2025-2032)
14.2. Asia Pacific Cell Culture Media Market Size and Forecast, by Form (2025-2032)
14.3. Asia Pacific Cell Culture Media Market Size and Forecast, by Application (2025-2032)
14.4. Asia Pacific Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
14.5. Asia Pacific Cell Culture Media Market Size and Forecast, by Country (2025-2032)
14.5.1. China
14.5.2. S Korea
14.5.3. Japan
14.5.4. India
14.5.5. Australia
14.5.6. Indonesia
14.5.7. Malaysia
14.5.8. Vietnam
14.5.9. Taiwan
14.5.10. Rest of Asia Pacific
15. Middle East and Africa Cell Culture Media Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
15.1. Middle East and Africa Cell Culture Media Market Size and Forecast, by Type (2025-2032)
15.2. Middle East and Africa Cell Culture Media Market Size and Forecast, by Form (2025-2032)
15.3. Middle East and Africa Cell Culture Media Market Size and Forecast, by Application (2025-2032)
15.4. Middle East and Africa Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
15.5. Middle East and Africa Cell Culture Media Market Size and Forecast, by Country (2025-2032)
15.5.1. South Africa
15.5.2. GCC
15.5.3. Egypt
15.5.4. Nigeria
15.5.5. Rest of ME&A
16. South America Cell Culture Media Market Size and Forecast by Segmentation (by Value USD Billion) (2025-2032)
16.1. South America Cell Culture Media Market Size and Forecast, by Type (2025-2032)
16.2. South America Cell Culture Media Market Size and Forecast, by Form (2025-2032)
16.3. South America Cell Culture Media Market Size and Forecast, by Application (2025-2032)
16.4. South America Cell Culture Media Market Size and Forecast, by End-Use (2025-2032)
16.5. South America Cell Culture Media Market Size and Forecast, by Country (2025-2032)
16.5.1. Brazil
16.5.2. Argentina
16.5.3. Chile
16.5.4. Colombia
16.5.5. Rest Of South America
17. Company Profile: Key Players
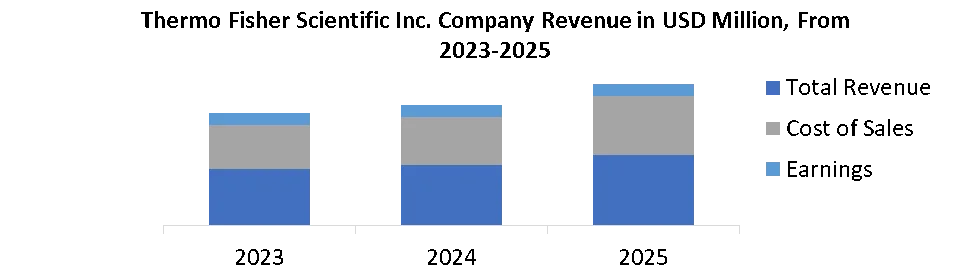
17.1. Thermo Fisher Scientific Inc.
17.1.1. Company Overview
17.1.2. Business Portfolio
17.1.3. Financial Overview
17.1.4. SWOT Analysis
17.1.5. Strategic Analysis
17.1.6. Recent Developments
17.2. Merck KGaA
17.3. Danaher Corporation (includes Cytiva)
17.4. Sartorius AG
17.5. Lonza Group AG
17.6. Corning Incorporated
17.7. Becton Dickinson (BD)
17.8. HiMedia Laboratories Pvt. Ltd.
17.9. Fujifilm Irvine Scientific (FUJIFILM)
17.10. Bio Techne Corporation
17.11. PromoCell GmbH
17.12. CellGenix GmbH
17.13. Takara Bio, Inc.
17.14. ZenBio, Inc.
17.15. Biological Industries Ltd.
17.16. Stemcell Technologies Inc.
17.17. Agilent Technologies, Inc.
17.18. Serumwerk Bernburg AG
17.19. Pan Biotech GmbH
17.20. Atlanta Biologicals, Inc.
17.21. Bio Rad Laboratories, Inc.
17.22. InVitria (Ventria Bioscience)
17.23. Allele Life Sciences (P) Ltd
17.24. PL BioScience GmbH
17.25. Alna Bioscience GmbH & Co. KG
17.26. Biomatik Corporation
17.27. Biologos
17.28. Creative Bioarray
17.29. Cell Culture Technologies
17.30. Quality Biological, Inc.AG
17.31. Others
18. Key Findings
19. Industry Recommendations
20. Cell Culture Media Market: Research Methodology
















