US Multiple Myeloma Therapeutics Market Industry Overview, Size, Share, Growth Trends, Research Insights and Forecast (2025–2032)
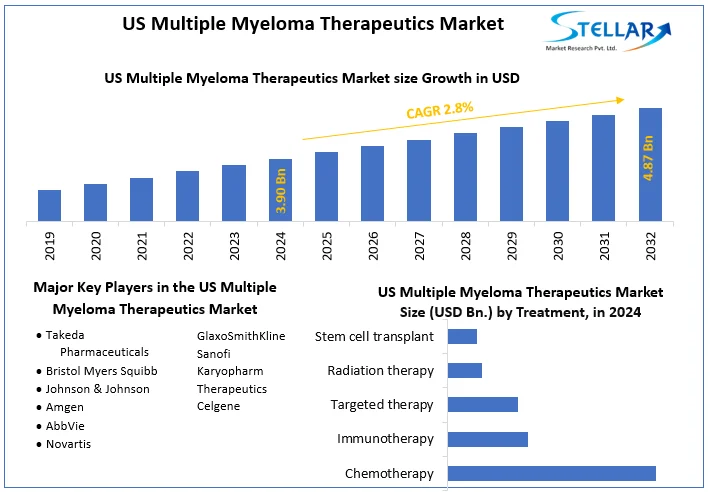
The US Multiple Myeloma Therapeutics Market size was valued at USD 3.90 Bn. in 2024 and the total US Multiple Myeloma Therapeutics revenue is expected to grow at a CAGR of 2.8 % from 2025 to 2032, reaching nearly USD 4.87 Bn. in 2032.
US Multiple Myeloma Therapeutics Market Overview-
Multiple myeloma (MM) is a challenging blood cancer marked by uncontrollable plasma cell proliferation in the bone marrow, producing non-functional proteins. Despite progress, Multiple myeloma remains mostly incurable, with standard frontline therapies providing limited, temporary relief. Drug resistance and genetic variations contribute to relapse. Treatment toxicity and refractoriness added limited options, necessitating innovative approaches against diverse myeloma cell populations. This overview addresses the current Multiple myeloma treatment landscape, emphasizing gaps in translating recent advances and exploring potential in targeted therapy and immunotherapy.
The comprehensive analysis of the US Multiple Myeloma Therapeutics market delves into trends, technological advancements, and potential disruptions, considering market size, growth, economic factors, regulations, and commercial drivers. The competitive landscape is examined, highlighting key operators' differentiation. Utilizing historical data, industry insights, and forecasts, the report addresses the sector's resilience challenges amid the economic downturn. Intended for healthcare professionals, government agencies, policymakers, and pharmaceutical companies, it aims to provide concise, crucial information for informed decision-making in this dynamic industry.
To get more Insights: Request Free Sample Report
Rising Incidence of Multiple Myeloma to Drive the US Multiple Myeloma Therapeutics Market
Growing multiple myeloma cases drive demand for effective treatments, presenting a US Multiple Myeloma Therapeutics market opportunity for pharmaceutical firms to develop new drugs. This leads to increased investment in research and development. Increased awareness raises early diagnoses, enabling timely interventions and potentially better patient outcomes, spurring demand for early-stage treatments.
In the US Multiple Myeloma Therapeutics Rising multiple myeloma cases strain healthcare budgets, burdening patients and systems. High therapy costs raise ethical concerns about equal access and disparities in healthcare outcomes among diverse demographics. Growing demand stresses resources, highlighting the need for efficient allocation and exploring alternative care models for equitable and adequate patient care.
The increasing number of multiple myeloma cases offers growth potential for the US Multiple Myeloma Therapeutics Market. However, it demands addressing challenges of healthcare affordability, equitable access, and resource management to sustain patient well-being and the healthcare system.
US Multiple Myeloma Therapeutics Market Segment Analysis
Based on the Distribution Channel, the Hospitals segment held the largest market share of about 65% in the US Multiple Myeloma Therapeutics Market in 2024. According to the STELLAR analysis, the segment is expected to grow at a CAGR of 2.8% during the forecast period. It stands out as the dominant segment within the US Multiple Myeloma Therapeutics Market thanks to its rapid technological advancement and growing adoption of smart devices with data connectivity and integration.
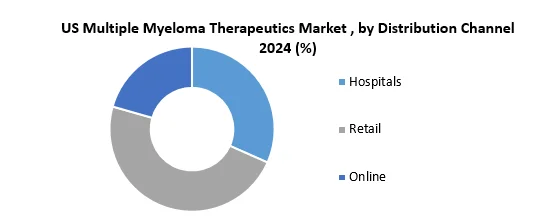
Hospitals play a crucial role in US multiple myeloma Therapeutics care, offering specialized expertise from hematologists and oncologists. They ensure the safe administration of involved treatments, vital for drugs requiring controlled settings. Hospitals provide vigilant monitoring, regular checkups, and blood tests, managing side effects. Participation in clinical trials widens treatment options, fostering innovation and improved patient outcomes.
The US Multiple myeloma therapeutics market poses financial challenges due to expensive hospital stays and treatments, burdening patients and healthcare systems. Limited access to specialized hospitals creates disparities, hindering equal healthcare outcomes. Overutilization of hospital resources may escalate costs, with some procedures suitable for outpatient clinics, highlighting potential inefficiencies in resource allocation.
Hospitals are crucial for the US multiple myeloma Therapeutics market, ensuring availability and proper administration. Addressing costs, enhancing access, and promoting efficient resource use is vital for a sustainable and equitable healthcare system.
US Multiple Myeloma Therapeutics Market Scope:
|
US Multiple Myeloma Therapeutics Market |
|
|
Market Size in 2024 |
USD 3.90 Billion |
|
Market Size in 2032 |
USD 4.87 Billion |
|
CAGR (2025-2032) |
2.8% |
|
Historic Data |
2019-2024 |
|
Base Year |
2024 |
|
Forecast Period |
2025-2032 |
|
|
By Treatment
|
|
By Distribution Channel
|
|
|
By End-User
|
|
Leading Key Players in the US Multiple Myeloma Therapeutics Market
- Takeda Pharmaceuticals
- Bristol Myers Squibb
- Johnson & Johnson
- Amgen
- AbbVie
- Novartis
- GlaxoSmithKline
- Sanofi
- Karyopharm Therapeutics
- Celgene
Frequently Asked Questions
High costs and Limited reimbursement are expected to be the major restraining factors for the US Multiple Myeloma Therapeutics market growth.
The US Multiple Myeloma Therapeutics Market size was valued at USD 3.90 Billion in 2024 and the total US Multiple Myeloma Therapeutics revenue is expected to grow at a CAGR of 2.8% from 2025 to 2032, reaching nearly USD 4.87 Billion By 2032.
1. US Multiple Myeloma Therapeutics Market Introduction
1.1 Study Assumption and Market Definition
1.2 Scope of the Study
1.3 Executive Summary
1.4 Emerging Technologies
1.5 Market Projections
1.6 Strategic Recommendations
2. US Multiple Myeloma Therapeutics Market Trends
2.1 Market Consolidation
2.2 Adoption of Advanced Technologies
2.3 Pricing and Reimbursement Trends
3. US Multiple Myeloma Therapeutics Market: Dynamics
3.1.1 Market Drivers
3.1.2 Market Restraints
3.1.3 Market Opportunities
3.1.4 Market Challenges
3.2 PORTER’s Five Forces Analysis
3.3 PESTLE Analysis
3.4 Regulatory Landscape
3.5 Analysis of Government Schemes and Initiatives for the US Multiple Myeloma Therapeutics Industry.
3.6 The Pandemic and Redefining of The US Multiple Myeloma Therapeutics Industry Landscape
4. US Multiple Myeloma Therapeutics Market: Market Size and Forecast by Segmentation (Value) (2024-2032)
4.1 US Multiple Myeloma Therapeutics Market Size and Forecast, By Treatment (2024-2032)
4.1.1 Chemotherapy
4.1.2 Immunotherapy
4.1.3 Targeted therapy
4.1.4 Radiation therapy
4.1.5 Stem cell transplant
4.2 US Multiple Myeloma Therapeutics Market Size and Forecast, By Distribution Channel (2024-2032)
4.2.1 Hospitals
4.2.2 Retail
4.2.3 Online
4.3 US Multiple Myeloma Therapeutics Market Size and Forecast, By End-user (2024-2032)
4.3.1 Hospitals
4.3.2 Clinics
4.3.3 Cancer Research Centers
5. US Multiple Myeloma Therapeutics Market: Competitive Landscape
5.1 STELLAR Competition Matrix
5.2 Competitive Landscape
5.3 Key Players Benchmarking
5.3.1 Company Name
5.3.2 Service Segment
5.3.3 End-user Segment
5.3.4 Revenue (2024)
5.3.5 Company Locations
5.4 Leading US Multiple Myeloma Therapeutics Companies, by Market Capitalization
5.5 Market Structure
5.5.1 Market Leaders
5.5.2 Market Followers
5.5.3 Emerging Players
5.6 Mergers and Acquisitions Details
6. Company Profile: Key Players
6.1 Takeda Pharmaceuticals
6.1.1 Company Overview
6.1.2 Business Portfolio
6.1.3 Financial Overview
6.1.4 SWOT Analysis
6.1.5 Strategic Analysis
6.1.6 Scale of Operation (small, medium, and large)
6.1.7 Details on Partnership
6.1.8 Regulatory Accreditations and Certifications Received by Them
6.1.9 Awards Received by the Firm
6.1.10 Recent Developments
6.2 Bristol Myers Squibb
6.3 Johnson & Johnson
6.4 Amgen
6.5 AbbVie
6.6 Novartis
6.7 GlaxoSmithKline
6.8 Sanofi
6.9 Karyopharm Therapeutics
6.10 Celgene
7. Key Findings
8. Industry Recommendations
9. US Multiple Myeloma Therapeutics Market: Research Methodology
















